£130.00
Retatrutide 30 mg (VioGen) delivers high‑strength triple‑agonist metabolic activation for advanced appetite control and accelerated fat‑loss performance.
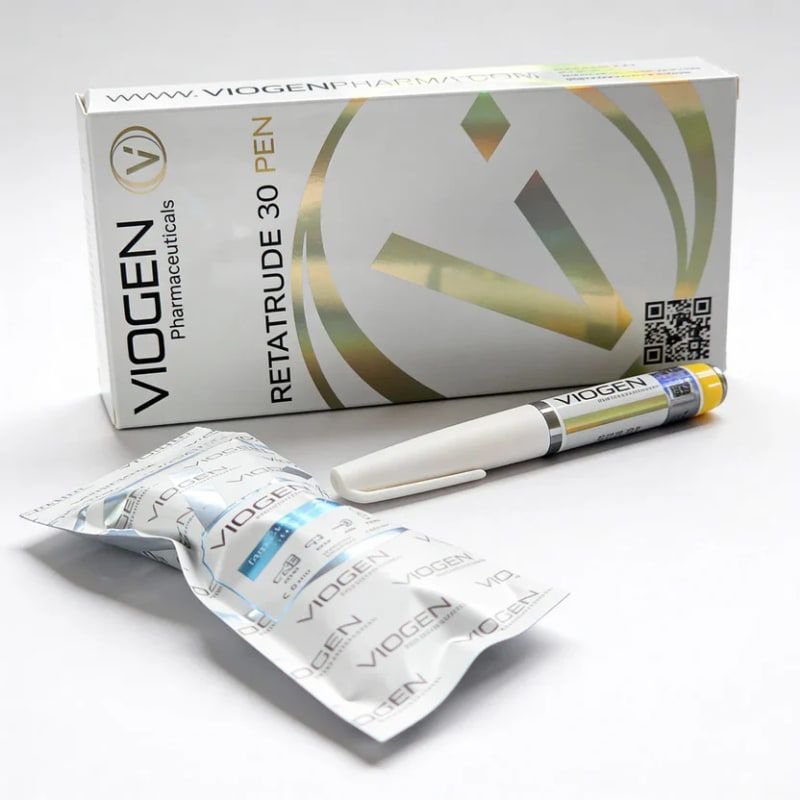
Buy Retatrutide 30mg (VioGen)
High‑Strength Triple‑Agonist Injection for Elite Metabolic Acceleration & Deep Fat‑Loss Performance
Retatrutide 30 mg (VioGen) delivers a powerful triple‑agonist formulation activating GIP, GLP‑1, and glucagon receptors simultaneously — the same mechanism behind Retatrutide’s record‑breaking clinical results, showing up to 24% body‑weight reduction at 48 weeks.
The 30 mg strength sits in the upper‑premium tier, offering stronger appetite suppression, deeper metabolic activation, and faster fat‑loss velocity than mid‑range doses. VioGen’s formulation is engineered for users seeking elite‑level metabolic performance without stepping into the ultra‑high 40 mg range.
Key Features
- High‑strength 30 mg triple‑agonist formulation
- Activates GIP + GLP‑1 + glucagon pathways
- Strong appetite suppression and craving reduction
- Accelerates fat oxidation and metabolic rate
- Supports rapid, visible fat‑loss progression
- Enhances insulin sensitivity and glycemic balance
- Once‑weekly injectable format
- Inspired by Retatrutide’s groundbreaking clinical outcomes
How Retatrutide 30 mg (VioGen) Works
GLP‑1 Activation
- Reduces appetite
- Slows gastric emptying
- Enhances satiety
GIP Activation
- Improves insulin response
- Supports metabolic stability
- Enhances tolerability
Glucagon Activation
- Increases energy expenditure
- Accelerates fat oxidation
- Drives deeper fat‑loss results
This triple‑pathway synergy produces superior metabolic efficiency and high‑velocity fat‑loss performance.
Benefits
- Strong appetite and craving control
- Faster fat‑loss progression
- Increased metabolic rate
- Improved glycemic balance
- Ideal for advanced users seeking high‑intensity metabolic support
- Once‑weekly convenience
Who It’s For
Retatrutide 30 mg (VioGen) is suited for:
- Users progressing from 10–20 mg triple‑agonist formulations
- Individuals requiring stronger appetite suppression
- Those targeting accelerated fat‑loss performance
- Users familiar with GLP‑1, GIP‑GLP‑1, or triple‑agonist peptides
Safety Information
Commonly reported effects:
- Mild nausea
- GI discomfort
- Fatigue
- Slight increase in heart rate (glucagon‑related)
Not suitable for:
- Pregnant or breastfeeding individuals
- Patients with personal/family history of medullary thyroid carcinoma
- Individuals with MEN2 syndrome
This product is not an approved pharmaceutical and is intended for research or professional use
Be the first to review “Buy Retatrutide 30mg” Cancel reply
Related products
Alluvi Brand
Alluvi Brand
VioGen
VioGen
Alluvi Brand
Alluvi Brand
Alluvi BPC-157 & TB-500 40mg
VioGen











Reviews
There are no reviews yet.